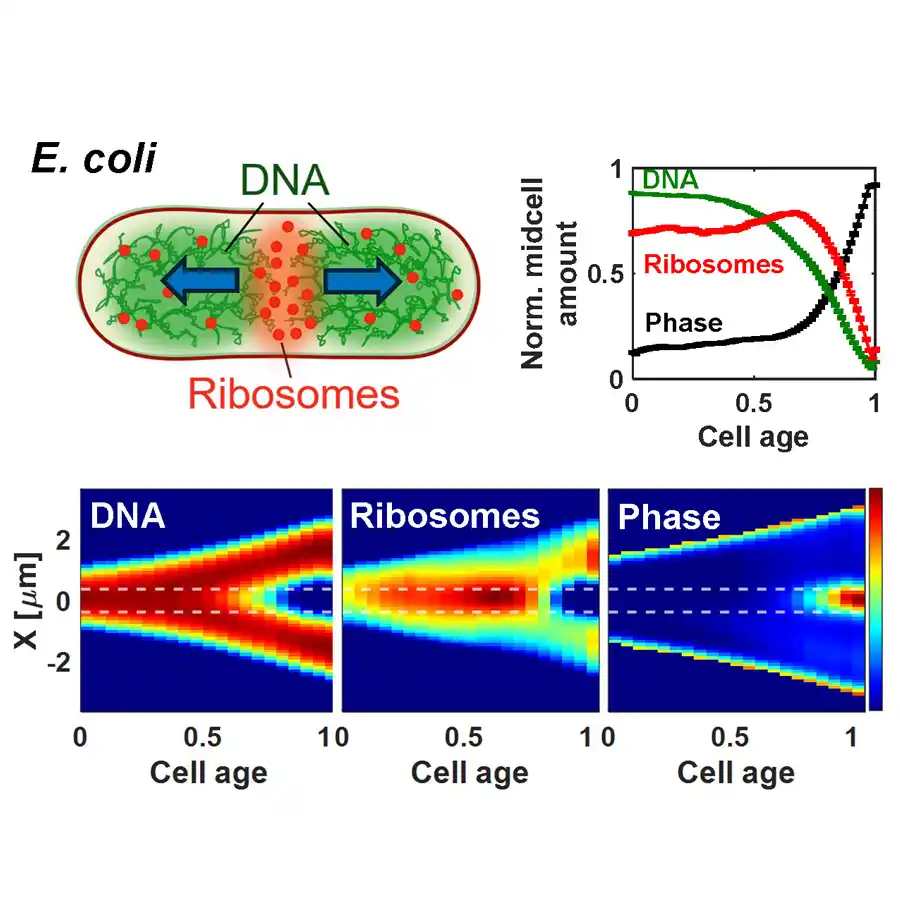
Physics After Dark

Signal:Noise welcomes a new DJ for their February 19 show!
Here’s the lineup:
- Science & Reason (DJ set): a mix of techno, dance, and house music (Bains Professor Steve Johnston)
- ColliderScope (VJ set): audio waveform-created images from CERN + sound waves across oscilloscope screens (Assistant Professor Larry Lee)
- Oskar (DJ set): Tennessee debut of Particle Physicist/DJ Oskar Hartbrich
Show begins at 9 p.m. (21+; No Cover) at Fly by Night